Xeikon white paper on migration-optimised digital printing solutions
The migration behaviour of digital ink systems plays a decisive role in the selection of a digital press and determines to a large extent which application segments the printing system can cover – and to which segments it could be expanded if necessary. These comprehensive aspects of migration-optimised solutions are discussed in Xeikon’s new white paper, in which Lode Deprez, vice-president, technology, provides insight into the criteria that must be taken into account when purchasing a digital printing system.
19 Feb 2021 | By PrintWeek Team
The white paper initially addresses the question of why there can’t be a “one ink” system that is suitable for all applications in digital packaging printing. It outlines the extremely complex requirements placed on ink systems, which differ in their migration behaviour and whose food safety properties are a very important criteria when selecting a machine system.
In addition, the ink systems used must meet legal requirements. The white paper provides a brief overview of the key regulations issued by legislative bodies, associations and branded goods manufacturers, and provides easy-to-understand information on the current migration limits.
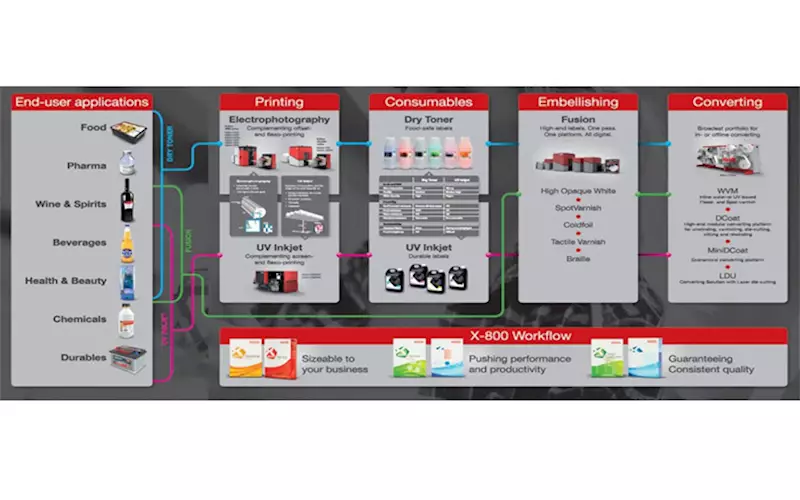
Against this background, it shows the importance of the chemical composition of digital ink systems on migration behaviour. By looking into the different composition of dry and liquid toner and UV inkjet inks, it highlights the advantages of the respective ink systems for different applications.
However, the ink components, with their different molecule sizes, are not the only elements to have an influence on the migration behaviour of digital ink systems. The transfer from printing inks to food depends on other factors such as storage time, storage temperature, barrier properties of the packaging material and, ultimately, the packaged good itself. The white paper also provides clear explanations on this topic.
The result is a clear picture of the user segments in which digital ink systems have their strengths. Divided into the segments of food, pharmaceuticals, wine and spirits, beverages, health and beauty, chemicals and consumer goods, individual recommendations can be derived for each label and packaging printer depending on the composition of their portfolio, which will help them when deciding on a digital printing system. The white paper also goes over the question of how to proceed if the portfolio is to be further developed in a particular direction.
Download the White Paper here.













 See All
See All